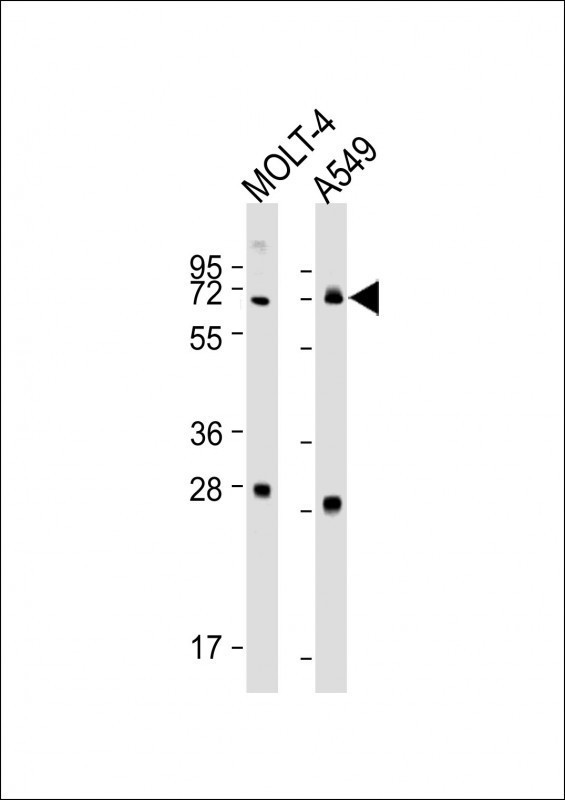
NEIL3 Antibody(Center)
Affinity Purified Rabbit Polyclonal Antibody (Pab)
- 产品详情
- 实验流程
- 背景知识
Application
| WB, E |
|---|---|
| Primary Accession | Q8TAT5 |
| Other Accession | NP_060718.2 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 67769 Da |
| Antigen Region | 292-320 aa |
| Gene ID | 55247 |
|---|---|
| Other Names | Endonuclease 8-like 3, 322-, DNA glycosylase FPG2, DNA glycosylase/AP lyase Neil3, Endonuclease VIII-like 3, Nei-like protein 3, NEIL3 |
| Target/Specificity | This NEIL3 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 292-320 amino acids from the Central region of human NEIL3. |
| Dilution | WB~~1:1000-1:2000 E~~Use at an assay dependent concentration. |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | NEIL3 Antibody(Center) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | NEIL3 |
|---|---|
| Function | DNA glycosylase which prefers single-stranded DNA (ssDNA), or partially ssDNA structures such as bubble and fork structures, to double-stranded DNA (dsDNA) (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). Mediates interstrand cross-link repair in response to replication stress: acts by mediating DNA glycosylase activity, cleaving one of the two N-glycosyl bonds comprising the interstrand cross-link, which avoids the formation of a double-strand break but generates an abasic site that is bypassed by translesion synthesis polymerases (By similarity). In vitro, displays strong glycosylase activity towards the hydantoin lesions spiroiminodihydantoin (Sp) and guanidinohydantoin (Gh) in both ssDNA and dsDNA; also recognizes FapyA, FapyG, 5-OHU, 5-OHC, 5-OHMH, Tg and 8-oxoA lesions in ssDNA (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). No activity on 8-oxoG detected (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). Also shows weak DNA-(apurinic or apyrimidinic site) lyase activity (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). In vivo, appears to be the primary enzyme involved in removing Sp and Gh from ssDNA in neonatal tissues (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). |
| Cellular Location | Nucleus. Chromosome {ECO:0000250|UniProtKB:A0A1L8HU22}. Note=Recruited to replication stress sites via interaction with ubiquitinated CMG helicase {ECO:0000250|UniProtKB:A0A1L8HU22} |
| Tissue Location | Expressed in keratinocytes and embryonic fibroblasts (at protein level). Also detected in thymus, testis and fetal lung primary fibroblasts. |
For Research Use Only. Not For Use In Diagnostic Procedures.
Provided below are standard protocols that you may find useful for product applications.
BACKGROUND
NEIL3 belongs to a class of DNA glycosylases homologous to the bacterial Fpg/Nei family. These glycosylases initiate the first step in base excision repair by cleaving bases damaged by reactive oxygen species and introducing a DNA strand break via the associated lyase reaction (Bandaru et al., 2002 [PubMed 12509226]).
REFERENCES
Krokeide, S.Z., et al. Protein Expr. Purif. 65(2):160-164(2009)
Takao, M., et al. Genes Cells 14(2):261-270(2009)
Dallosso, A.R., et al. Gut 57(9):1252-1255(2008)
Bethke, L., et al. J. Natl. Cancer Inst. 100(4):270-276(2008)
Newton-Cheh, C., et al. BMC Med. Genet. 8 SUPPL 1, S7 (2007) :
终于等到您。ABCEPTA(百远生物)抗体产品。
点击下方“我要评价 ”按钮提交您的反馈信息,您的反馈和评价是我们最宝贵的财富之一,
我们将在1-3个工作日内处理您的反馈信息。
如有疑问,联系:0512-88856768 tech-china@abcepta.com.























 癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。
癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。 为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。
为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。 细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。
细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。