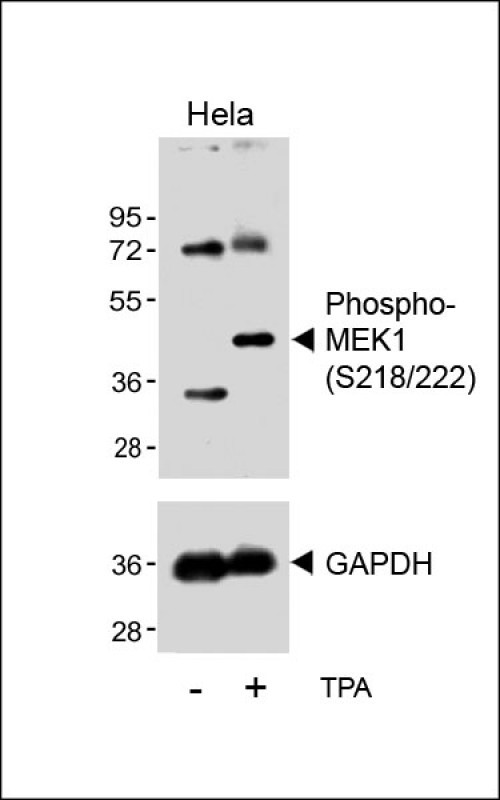
Bi-Phospho-MEK1(S218/222) Antibody
Affinity Purified Rabbit Polyclonal Antibody (Pab)
- 产品详情
- 实验流程
- 背景知识
Application
| WB, IHC-P, DB, E |
|---|---|
| Primary Accession | Q02750 |
| Other Accession | P36506, Q63932, P36507, Q90891, Q05116, Q01986, P29678, P31938, Q63980, Q10664, Q24324 |
| Reactivity | Human |
| Predicted | Mouse, Rat, Rabbit, Hamster, Chicken, Drosophila, C.Elegans, Xenopus |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 43439 Da |
| Gene ID | 5604 |
|---|---|
| Other Names | Dual specificity mitogen-activated protein kinase kinase 1, MAP kinase kinase 1, MAPKK 1, MKK1, ERK activator kinase 1, MAPK/ERK kinase 1, MEK 1, MAP2K1, MEK1, PRKMK1 |
| Target/Specificity | This MEK1 Antibody is generated from rabbits immunized with a KLH conjugated synthetic phosphopeptide corresponding to amino acid residues surrounding S218/222 of human MEK1. |
| Dilution | WB~~1:500 IHC-P~~1:100~500 DB~~1:500 E~~Use at an assay dependent concentration. |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | Bi-Phospho-MEK1(S218/222) Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | MAP2K1 (HGNC:6840) |
|---|---|
| Synonyms | MEK1, PRKMK1 |
| Function | Dual specificity protein kinase which acts as an essential component of the MAP kinase signal transduction pathway. Binding of extracellular ligands such as growth factors, cytokines and hormones to their cell-surface receptors activates RAS and this initiates RAF1 activation. RAF1 then further activates the dual-specificity protein kinases MAP2K1/MEK1 and MAP2K2/MEK2. Both MAP2K1/MEK1 and MAP2K2/MEK2 function specifically in the MAPK/ERK cascade, and catalyze the concomitant phosphorylation of a threonine and a tyrosine residue in a Thr-Glu-Tyr sequence located in the extracellular signal-regulated kinases MAPK3/ERK1 and MAPK1/ERK2, leading to their activation and further transduction of the signal within the MAPK/ERK cascade. Activates BRAF in a KSR1 or KSR2-dependent manner; by binding to KSR1 or KSR2 releases the inhibitory intramolecular interaction between KSR1 or KSR2 protein kinase and N-terminal domains which promotes KSR1 or KSR2-BRAF dimerization and BRAF activation (PubMed:29433126). Depending on the cellular context, this pathway mediates diverse biological functions such as cell growth, adhesion, survival and differentiation, predominantly through the regulation of transcription, metabolism and cytoskeletal rearrangements. One target of the MAPK/ERK cascade is peroxisome proliferator-activated receptor gamma (PPARG), a nuclear receptor that promotes differentiation and apoptosis. MAP2K1/MEK1 has been shown to export PPARG from the nucleus. The MAPK/ERK cascade is also involved in the regulation of endosomal dynamics, including lysosome processing and endosome cycling through the perinuclear recycling compartment (PNRC), as well as in the fragmentation of the Golgi apparatus during mitosis. |
| Cellular Location | Cytoplasm, cytoskeleton, microtubule organizing center, centrosome. Cytoplasm, cytoskeleton, microtubule organizing center, spindle pole body. Cytoplasm. Nucleus Membrane; Peripheral membrane protein. Note=Localizes at centrosomes during prometaphase, midzone during anaphase and midbody during telophase/cytokinesis (PubMed:14737111). Membrane localization is probably regulated by its interaction with KSR1 (PubMed:10409742) |
| Tissue Location | Widely expressed, with extremely low levels in brain. |
For Research Use Only. Not For Use In Diagnostic Procedures.
Provided below are standard protocols that you may find useful for product applications.
BACKGROUND
MEK1is a dual specificity protein kinase that belongs to the MAP kinase kinase family. This kinase is known to play a critical role in mitogen growth factor signal transduction. It phosphorylates and thus activates MAPK1/ERK2 and MAPK2/ERK3. The activation of this kinase itself is dependent on the Ser/Thr phosphorylation by MAP kinase kinase kinases. The inhibition or degradation of this kinase is found to be involved in the pathogenesis of Yersinia and anthrax.
REFERENCES
Ohren, J.F., et al., J. Neural Transm. 11(12):1192-1197 (2004). Naegele, S., et al., J. Biol. Chem. 279(44):46023-46034 (2004). Ussar, S., et al., J. Biol. Chem. 279(42):43861-43869 (2004). Beausoleil, S.A., et al., Proc. Natl. Acad. Sci. U.S.A. 101(33):12130-12135 (2004). Spence, H.J., et al., EMBO Rep. 5(5):484-489 (2004).
终于等到您。ABCEPTA(百远生物)抗体产品。
点击下方“我要评价 ”按钮提交您的反馈信息,您的反馈和评价是我们最宝贵的财富之一,
我们将在1-3个工作日内处理您的反馈信息。
如有疑问,联系:0512-88856768 tech-china@abcepta.com.























 癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。
癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。 为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。
为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。 细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。
细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。