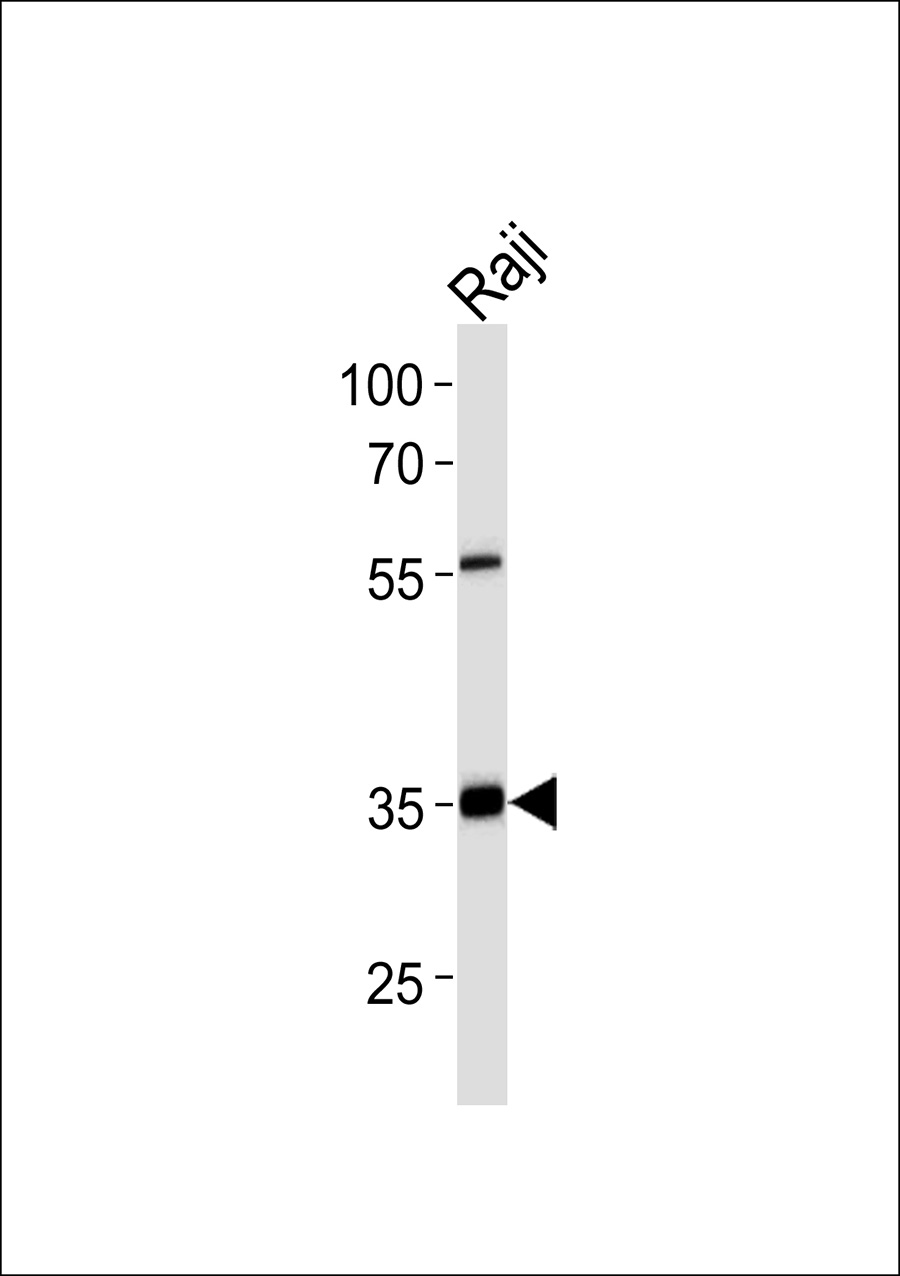
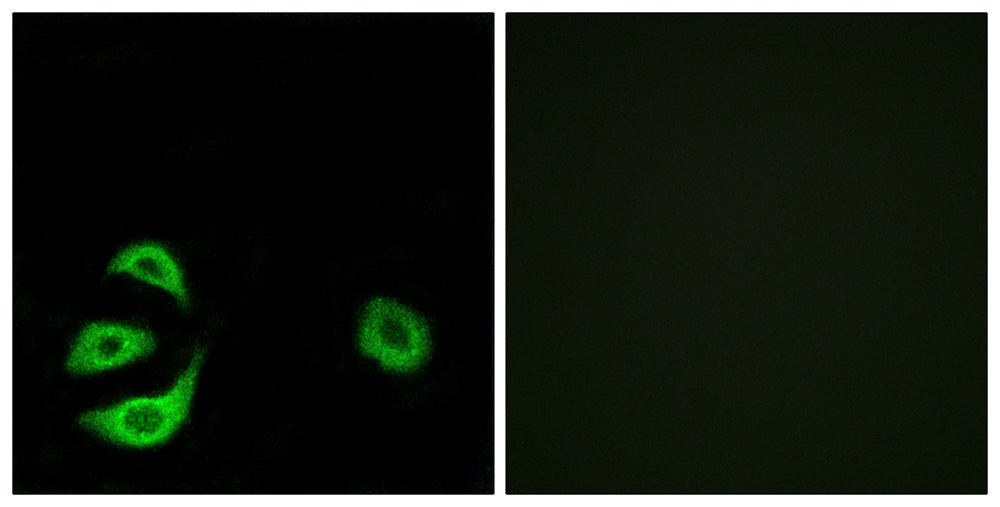
ACOT8 Antibody
Purified Rabbit Polyclonal Antibody (Pab)
- 产品详情
- 实验流程
- 背景知识
Application
| WB, IF |
|---|---|
| Primary Accession | O14734 |
| Reactivity | Human, Mouse, Rat |
| Host | Rabbit |
| Clonality | polyclonal |
| Calculated MW | 35914 Da |
| Gene ID | 10005 |
|---|---|
| Other Names | Acyl-coenzyme A thioesterase 8, Acyl-CoA thioesterase 8, Choloyl-coenzyme A thioesterase, HIV-Nef-associated acyl-CoA thioesterase, PTE-2, Peroxisomal acyl-coenzyme A thioester hydrolase 1, PTE-1, Peroxisomal long-chain acyl-CoA thioesterase 1, Thioesterase II, hACTE-III, hACTEIII, hTE, ACOT8, ACTEIII, PTE1, PTE2 |
| Dilution | WB~~ 1:1000 IF~~1:100 |
| Format | Rabbit IgG in phosphate buffered saline (without Mg2+ and Ca2+), pH 7.4, 150mM NaCl, 0.09% (W/V) sodium azide and 50% glycerol. |
| Storage Conditions | -20℃ |
| Name | ACOT8 |
|---|---|
| Synonyms | ACTEIII, PTE1 {ECO:0000303|PubMed:100925 |
| Function | Catalyzes the hydrolysis of acyl-CoAs into free fatty acids and coenzyme A (CoASH), regulating their respective intracellular levels (PubMed:15194431, PubMed:9153233, PubMed:9299485). Displays no strong substrate specificity with respect to the carboxylic acid moiety of Acyl-CoAs (By similarity). Hydrolyzes medium length (C2 to C20) straight-chain, saturated and unsaturated acyl-CoAS but is inactive towards substrates with longer aliphatic chains (PubMed:9153233, PubMed:9299485). Moreover, it catalyzes the hydrolysis of CoA esters of bile acids, such as choloyl-CoA and chenodeoxycholoyl-CoA and competes with bile acid CoA:amino acid N-acyltransferase (BAAT) (By similarity). Is also able to hydrolyze CoA esters of dicarboxylic acids (By similarity). It is involved in the metabolic regulation of peroxisome proliferation (PubMed:15194431). |
| Cellular Location | Peroxisome matrix. Note=Predominantly localized in the peroxisome but a localization to the cytosol cannot be excluded |
| Tissue Location | Detected in a T-cell line (at protein level). Ubiquitous (PubMed:9153233, PubMed:9299485) |
For Research Use Only. Not For Use In Diagnostic Procedures.
Provided below are standard protocols that you may find useful for product applications.
BACKGROUND
Acyl-CoA thioesterases are a group of enzymes that catalyze the hydrolysis of acyl-CoAs to the free fatty acid and coenzyme A (CoASH), providing the potential to regulate intracellular levels of acyl-CoAs, free fatty acids and CoASH. May mediate Nef-induced down-regulation of CD4. Major thioesterase in peroxisomes. Competes with BAAT (Bile acid CoA: amino acid N- acyltransferase) for bile acid-CoA substrate (such as chenodeoxycholoyl-CoA). Shows a preference for medium-length fatty acyl-CoAs (By similarity). May be involved in the metabolic regulation of peroxisome proliferation.
REFERENCES
Watanabe H.,et al.Biochem. Biophys. Res. Commun. 238:234-239(1997).
Liu L.X.,et al.J. Biol. Chem. 272:13779-13785(1997).
Jones J.M.,et al.J. Biol. Chem. 274:9216-9223(1999).
Deloukas P.,et al.Nature 414:865-871(2001).
Ishizuka M.,et al.Exp. Cell Res. 297:127-141(2004).
终于等到您。ABCEPTA(百远生物)抗体产品。
点击下方“我要评价 ”按钮提交您的反馈信息,您的反馈和评价是我们最宝贵的财富之一,
我们将在1-3个工作日内处理您的反馈信息。
如有疑问,联系:0512-88856768 tech-china@abcepta.com.























 癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。
癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。 为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。
为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。 细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。
细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。