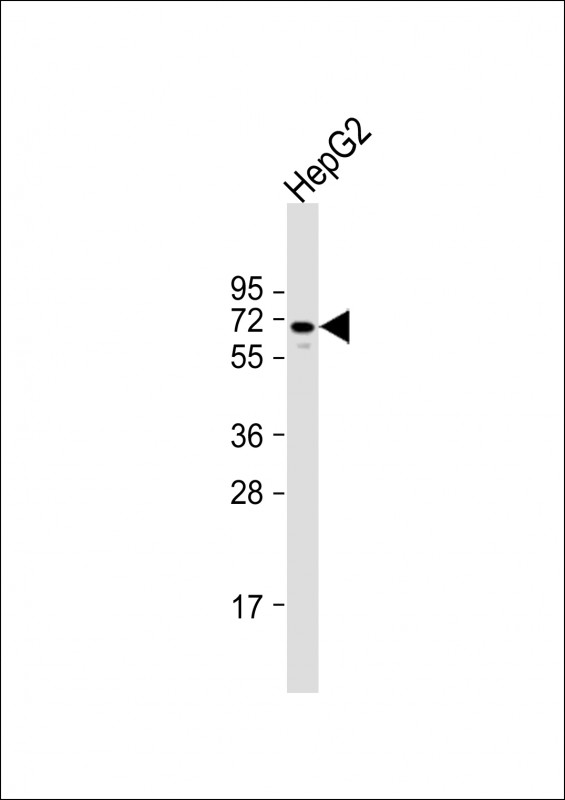
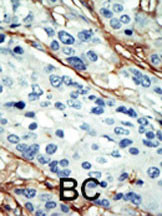
ARK5 Antibody (C-term)
Purified Rabbit Polyclonal Antibody (Pab)
- 产品详情
- 文献引用 : 1
- 实验流程
- 背景知识
Application
| WB, IHC-P, E |
|---|---|
| Primary Accession | O60285 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 74305 Da |
| Antigen Region | 604-634 aa |
| Gene ID | 9891 |
|---|---|
| Other Names | NUAK family SNF1-like kinase 1, AMPK-related protein kinase 5, ARK5, Omphalocele kinase 1, NUAK1, ARK5, KIAA0537, OMPHK1 |
| Target/Specificity | This ARK5 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 604-634 amino acids from the C-terminal region of human ARK5. |
| Dilution | WB~~1:1000 IHC-P~~1:100~500 E~~Use at an assay dependent concentration. |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | ARK5 Antibody (C-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | NUAK1 |
|---|---|
| Synonyms | ARK5, KIAA0537, OMPHK1 |
| Function | Serine/threonine-protein kinase involved in various processes such as cell adhesion, regulation of cell ploidy and senescence, cell proliferation and tumor progression. Phosphorylates ATM, CASP6, LATS1, PPP1R12A and p53/TP53. Acts as a regulator of cellular senescence and cellular ploidy by mediating phosphorylation of 'Ser-464' of LATS1, thereby controlling its stability. Controls cell adhesion by regulating activity of the myosin protein phosphatase 1 (PP1) complex. Acts by mediating phosphorylation of PPP1R12A subunit of myosin PP1: phosphorylated PPP1R12A then interacts with 14-3-3, leading to reduced dephosphorylation of myosin MLC2 by myosin PP1. May be involved in DNA damage response: phosphorylates p53/TP53 at 'Ser-15' and 'Ser-392' and is recruited to the CDKN1A/WAF1 promoter to participate in transcription activation by p53/TP53. May also act as a tumor malignancy-associated factor by promoting tumor invasion and metastasis under regulation and phosphorylation by AKT1. Suppresses Fas-induced apoptosis by mediating phosphorylation of CASP6, thereby suppressing the activation of the caspase and the subsequent cleavage of CFLAR. Regulates UV radiation-induced DNA damage response mediated by CDKN1A. In association with STK11, phosphorylates CDKN1A in response to UV radiation and contributes to its degradation which is necessary for optimal DNA repair (PubMed:25329316). |
| Cellular Location | Nucleus. Cytoplasm |
| Tissue Location | Expressed at high levels in heart and brain, and at lower levels in skeletal muscle, kidney, ovary, placenta, lung and liver. Highly up-regulated in colorectal cancer cell lines |
For Research Use Only. Not For Use In Diagnostic Procedures.

Provided below are standard protocols that you may find useful for product applications.
BACKGROUND
Protein kinases are enzymes that transfer a phosphate group from a phosphate donor, generally the g phosphate of ATP, onto an acceptor amino acid in a substrate protein. By this basic mechanism, protein kinases mediate most of the signal transduction in eukaryotic cells, regulating cellular metabolism, transcription, cell cycle progression, cytoskeletal rearrangement and cell movement, apoptosis, and differentiation. With more than 500 gene products, the protein kinase family is one of the largest families of proteins in eukaryotes. The family has been classified in 8 major groups based on sequence comparison of their tyrosine (PTK) or serine/threonine (STK) kinase catalytic domains. The STE group (homologs of yeast Sterile 7, 11, 20 kinases) consists of 50 kinases related to the mitogen-activated protein kinase (MAPK) cascade families (Ste7/MAP2K, Ste11/MAP3K, and Ste20/MAP4K). MAP kinase cascades, consisting of a MAPK and one or more upstream regulatory kinases (MAPKKs) have been best characterized in the yeast pheromone response pathway. Pheromones bind to Ste cell surface receptors and activate yeast MAPK pathway.
REFERENCES
Suzuki, A., et al., Mol. Cell. Biol. 24(8):3526-3535 (2004).
Suzuki, A., et al., Oncogene 22(40):6177-6182 (2003).
Suzuki, A., et al., J. Biol. Chem. 278(1):48-53 (2003).
终于等到您。ABCEPTA(百远生物)抗体产品。
点击下方“我要评价 ”按钮提交您的反馈信息,您的反馈和评价是我们最宝贵的财富之一,
我们将在1-3个工作日内处理您的反馈信息。
如有疑问,联系:0512-88856768 tech-china@abcepta.com.






















 癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。
癌症的基本特征包括细胞增殖、血管生成、迁移、凋亡逃避机制和细胞永生等。找到癌症发生过程中这些通路的关键标记物和对应的抗体用于检测至关重要。 为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。
为您推荐一个泛素化位点预测神器——泛素化分析工具,可以为您的蛋白的泛素化位点作出预测和评分。 细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。
细胞自噬受体图形绘图工具为你的蛋白的细胞受体结合位点作出预测和评分,识别结合到自噬通路中的蛋白是非常重要的,便于让我们理解自噬在正常生理、病理过程中的作用,如发育、细胞分化、神经退化性疾病、压力条件下、感染和癌症。